Newsletters

Nephrodite Announces Industry’s First Successful Multi-Day Large Animal Study for a Fully Implantable Renal Replacement Device
October 30, 2025
No Comments
The Holly™ by Nephrodite device demonstrated continuous kidney function replacement in a large animal marking a critical milestone toward commercialization
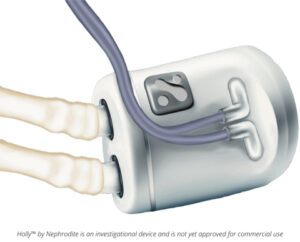
Nephrodite Earns FDA’s First Breakthrough Device Designation for an Implantable Kidney Replacement System
October 30, 2025
No Comments
Holly™ by Nephrodite becomes the first device to achieve the designation, marking a major regulatory milestone toward first-in-human clinical studies
Continuous dialysis implant wins FDA Breakthrough Device Designation
October 28, 2025
1 Comment
The FDA has granted Breakthrough Device Designation to Nephrodite’s Holly implantable continuous dialysis system. In an exclusive interview ahead
